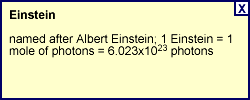Guide to UV Measurement
UV Basics
The electromagnetic spectrum spans a broad range of wavelengths, from long wavelength, low energy radio waves with a wavelength of a meter or more to short wavelength, high energy X-rays with a wavelength of less than one-billionth of a meter. The ultraviolet (UV) and visible regions comprise a small part of the total energy spectrum.

The energy E of a photon of any given wavelength ( λ ) is defined by the relationship
![]()
where h is Planck's constant and v is the frequency of the electromagnetic wave defined by c = λv in which c is the speed of light.
Visible light falls in the region from about 400-700 nanometers (abbreviated nm; 1 nm is equal to 10-9 or one-billionth of a meter). An Einstein of red light at 630 nm carries slightly more than 190 kilojoules (kJ) of energy while an Einstein at 400 nm carries about 300 kJ of energy. This amount of energy is below the typical bond strengths of most organic molecules and is therefore insufficient to break chemical bonds.
The ultraviolet region of the spectrum starts around 400 nm at the blue end of the visible spectrum and extends to about 100 nm. For purposes of conventional UV curing processes, the effective UV wavelength cut-off point is closer to 200 nm; below this, air becomes an efficient absorber of the energy. In the region from 200-400 nm, the amount of energy carried by photons ranges from about 300 to 600 kJ/Einstein which is comparable to the bond strengths of many organic molecules. Hence, when a molecule absorbs energy from photons in the ultraviolet wavelength region, chemical bonds within the molecular structure can be weakened or broken. This is the basis of all UV curing processes. To learn more, click here.